
A catalyst
changes the rate of a reaction without appearing in the products and without
changing the chemical equilibrium. A catalyst is a compound that lowers(or
higher) the free activation enthalpy energy of the reaction.

The term photocatalysts consists of the combination
of photochemisty and catalysis and impies that light and a catalyst are
necessary to bring about or to accelerate a chemical
transformation.
Photocatalyst can be induced photoreaction by the presence of
a catalyst with the light with various photocatalysts, the titanum dioxide is
wide used because it has stable about acid-base condition and harmless about
human body. 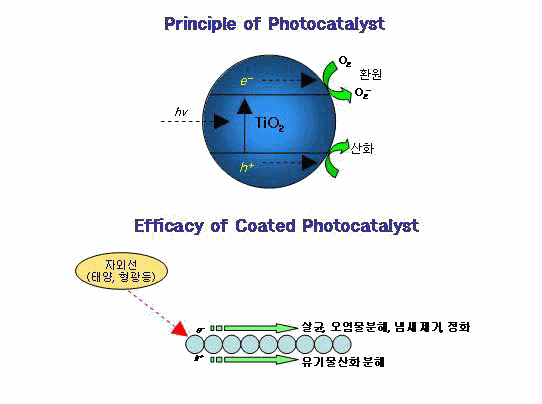

Titan is the 9th-most element on earth and titanium oxide is usually used
to white pigment and white paint for cosmetics,For pigments, no response to
lighrt-titanium oxide is used while for photocatalyst applications like air-purifiers,
high photo reactive titanium oxide is used. And due to its low price, excellent
wear-resistance, endurance ane no possibility of the secondary contamination
for its abundance, it has safe and nonpoisonous feature. So titanium oxide is
considered one of the most popular NANO products in future-oriented industry.
In particular, given the conditions and activeness of photocatalyst, it can
be used semi-permanently, since it doesn't change itself even when a light hits.
And its higher oxidation than CI2 and O3,excellent sterilization and function
of decomposing all organic compounds into H2O and CO2 allow TiO2 to be one of
the most popular photocatalyst materials.

 Safety of TiO2 Safety of TiO2
TiO2 used as photocatalyst is physically and chemically safe, the US certified
it as food additions in 1968 and Japan did in 1983 so that it is used widely
in making white chocolates and functional cosmetics including lipsticks. Particularly
in Japan, company T produced photocatalyst-coated tiles after Food Analysis
Center of Japan Food Research Laboratories and Mitsubishi Chemical Safety Institute
Ltd. demonstrated its safety through skin simulating tests and acute oral toxicity
test.
 Safety of Activated Oxygen Safety of Activated Oxygen
Active oxygen always exists in the air and it is created in human body. And
massive active oxygen can do harm to human body. Active oxygen created by photocatalyst
is always on the surface of coated materials, not in the air. And H2O2
is a good example to be used in daily lives. H2O2 sterilizes bacteria and other
germs by creating active oxygen on human skins.
|